 Amid the Zeppelin designs created after the Great War, after the Lecarno Treaties and even after LZ-127 was built, one of the most persistent myths in LTA has LZ-129 originally having been designed for pure helium lift. Some authors insist its increased size is a direct result of compensating for helium’s lesser lift. This miss-information is often piled on with some gifted hindsight that American officials refused sale of the gas because somehow in the early 1930s they foresaw Hitler’s threat and imagined he’d use helium for war purposes.
Amid the Zeppelin designs created after the Great War, after the Lecarno Treaties and even after LZ-127 was built, one of the most persistent myths in LTA has LZ-129 originally having been designed for pure helium lift. Some authors insist its increased size is a direct result of compensating for helium’s lesser lift. This miss-information is often piled on with some gifted hindsight that American officials refused sale of the gas because somehow in the early 1930s they foresaw Hitler’s threat and imagined he’d use helium for war purposes.
 True, the National Socialists did eventually come to the conclusion Zeppelins would be a unique, visible and profitable asset to the Reich. A memo from the propaganda minister for Adolph Hitler (later uncovered by Prof. Henry Meyer) urged the Zeppelin construction and operations be nationalized. Hitler okayed the plan, which would infuse cash to speed construction of the previously slow-moving LZ-129 project. Previous plans that never left the drawing board – or model form (photo) now seemed possible. These new dreams did not include the prohibitively expensive infrastructure for importing, processing and switching over to helium gas for lift.
True, the National Socialists did eventually come to the conclusion Zeppelins would be a unique, visible and profitable asset to the Reich. A memo from the propaganda minister for Adolph Hitler (later uncovered by Prof. Henry Meyer) urged the Zeppelin construction and operations be nationalized. Hitler okayed the plan, which would infuse cash to speed construction of the previously slow-moving LZ-129 project. Previous plans that never left the drawing board – or model form (photo) now seemed possible. These new dreams did not include the prohibitively expensive infrastructure for importing, processing and switching over to helium gas for lift.
In fact, evidence suggests the R-101 accident caused the Zeppelin men to scrap their plans for an “enlarged Graf,” the LZ-128, within a year of that tragedy.  The Graf’s Captain, Hans Von Schiller, wrote in The Airship Experience, “With the Hindenburg, the principle of gasoline and vapor motors was abandoned, and only diesel engines were used. Does this mean that gaseous fuel has not proved satisfactory? It does not. Properly, gas is still the ideal fuel from the technical point of view… As with the use of liquid fuel each kilogram of gasoline or oil costs one cubic meter of hydrogen to restore the equilibrium…”
The Graf’s Captain, Hans Von Schiller, wrote in The Airship Experience, “With the Hindenburg, the principle of gasoline and vapor motors was abandoned, and only diesel engines were used. Does this mean that gaseous fuel has not proved satisfactory? It does not. Properly, gas is still the ideal fuel from the technical point of view… As with the use of liquid fuel each kilogram of gasoline or oil costs one cubic meter of hydrogen to restore the equilibrium…”
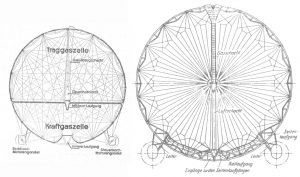 In the graphic here (not to scale) one can see a rather large volume of the Graf was taken up by its fuel gas. Since efficiency is all about lifting BTUs needed for propulsion energy, one understands why the extra volume of hydrogen needed to lift tonnage of diesel oil (whose weight then disappeared) was not more efficient than using weightless fuel. When one added up the weighty structure needed for the pinpoint loads of diesel fuel tanks, the rainwater weight-recovery guttering and tankage, etc., one can see why Von Schiller saw LZ-129’s adoption of diesel oil as a step backward. As C.P. Burgess pointed out, gaseous fuel would be most useful on a helium-filled airship. However, no plan for LZ-129 to be helium-lift/blau powered has ever been found. In fact, only one helium airship, the non-rigid K-1, even tried the ideal “weightless” fuel.
In the graphic here (not to scale) one can see a rather large volume of the Graf was taken up by its fuel gas. Since efficiency is all about lifting BTUs needed for propulsion energy, one understands why the extra volume of hydrogen needed to lift tonnage of diesel oil (whose weight then disappeared) was not more efficient than using weightless fuel. When one added up the weighty structure needed for the pinpoint loads of diesel fuel tanks, the rainwater weight-recovery guttering and tankage, etc., one can see why Von Schiller saw LZ-129’s adoption of diesel oil as a step backward. As C.P. Burgess pointed out, gaseous fuel would be most useful on a helium-filled airship. However, no plan for LZ-129 to be helium-lift/blau powered has ever been found. In fact, only one helium airship, the non-rigid K-1, even tried the ideal “weightless” fuel.  This in spite of a fuel gas plant having been built at Lakehurst, as seen in this photo.
This in spite of a fuel gas plant having been built at Lakehurst, as seen in this photo.
Nor had the Germans then begun any kind of an effort to import and handle the helium gas. As Professor Henry Meyer states, “Still, there is a remarkable ambiguity about [Eckener’s] forcefulness of his search for the essential nonflammable gas… To date there has appeared no document recording a formal application for helium by Eckener, or by any other German authorities prior to May 6, 1937.” For those who insist Prof. Meyer somehow missed what surely would be a most certain desire to have fire-safe gas, one only realize there was hardly any point in asking for a price quote – since at the time there was no actual method of moving large quantities of helium across the Atlantic. Indeed, at the height of World War II, the US Navy was still trying to support a few blimps in the Med with K-bottles of helium – about 210 cu ft each.
In the aftermath of the horror of R-101’s unidentifiable victim’s remains buried en mass in a Cardington sepulcher, improved safety was obviously paramount. Thankfully few claimed the British had secretly wanted helium, because in fact they had rejected an offer of free helium in exchange for expertise years earlier. It is too bad the gasoline-atop-diesel was not recognized as the only item present in R.101 that could have rendered the human bodies so indistinguishable. Still, since the new Mybach diesels did not need the gasoline starter motors like R.101, the diesel oil alone had a higher (safer) ignition temperature. To address the lifting gas safety issue, on the wish list was one emerging design that sought to use a sort of helium-wrap cell around the disposable-lift hydrogen cells.

Though not detailed outside of Germany, it was no secret, and in fact popular media concluded the ship had actually been operated that way. A 1936 photo labeled “Inside A Zeppelin” released by Bruno & Associates has the caption that read “…There are sixteen gas cells in the hull … The gas cells have a hydrogen center enclosed by helium.” Others slurred this to mean the rare gas was some sort of option. The U.S. Air Services magazine echoed that April, “The gas cells of the Hindenburg are made to take hydrogen, but can be changed to use both hydrogen and helium. As the ship goes into service hydrogen filled cells will be enclosed in larger helium-filled bags, additional hydrogen to be supplied at Lakehurst terminal from the Bayway plant of the Standard Oil Company of New Jersey under special arrangement with the Zeppelin operators.” Just as people swear they “heard” Herb Morrison’s “radio broadcast” the night of May 6th, 1937, one can find people who know for a fact LZ-129 would have been helium-filled if only the Americans had offered helium, cheap.
It simply isn’t so, no matter how many times it is repeated on TV.
As Laurie Soffe explained in Gasbag Journal #44, “In 1934, with the framework virtually complete, an actual trial was made in the ship with a pair of main cells inside which were made from the old cells from the LZ-127 (nearly six years old by then). Hydrogen was used in both cells for the trials, which although they were considered to be largely satisfactory, did not convince those who would be actually flying the ship. Their view was that with the long and successful experience built up with hydrogen, they should stick with it [only], and that view eventually prevailed.” Today, this is the only drawing of the original “wish list” design option found in the Zeppelin Museum.
 The literature also suggests study showed the different gasses would not fill and shape the cells equally, putting undue strain on the structure. Goodyear-Zeppelin’s Litchfield and Allen would publish, “The Zeppelin Captains wanted to go ahead with [only] hydrogen. All their flying for 25 years, in peace and war, had been with hydrogen. Their Zeppelins were expertly bonded against electrical disturbances, and hydrogen airships had flow safely though lightning storms many times. The crews wore felt shoes and buttonless jackets to avoid creating static electricity. Operated carefully, hydrogen was just as safe as helium, had better lift, was cheaper, [and] could be made anywhere. They did not need to ask the favor of helium from America.” Nonetheless the myth of German desire for helium in the early 1930s remains second only to the popular misconception the Hindenburg’s hydrogen exploded.
The literature also suggests study showed the different gasses would not fill and shape the cells equally, putting undue strain on the structure. Goodyear-Zeppelin’s Litchfield and Allen would publish, “The Zeppelin Captains wanted to go ahead with [only] hydrogen. All their flying for 25 years, in peace and war, had been with hydrogen. Their Zeppelins were expertly bonded against electrical disturbances, and hydrogen airships had flow safely though lightning storms many times. The crews wore felt shoes and buttonless jackets to avoid creating static electricity. Operated carefully, hydrogen was just as safe as helium, had better lift, was cheaper, [and] could be made anywhere. They did not need to ask the favor of helium from America.” Nonetheless the myth of German desire for helium in the early 1930s remains second only to the popular misconception the Hindenburg’s hydrogen exploded.
 The Zeppelin men also took note of another lesson learned by the British rigid program: aluminum alloys corrode. Images of the “rotten” R-100 girders being broken up and sold for scrap undoubtedly inspired the LZ-129 structure’s treatment with alkyd/acrylic lacquer containing a phthalocynanine blue dye. (Photo: This was accurately formulated for priming every piece of the Zeppelin Museum’s LZ-129 section re-creation.) The fact it was still flammable after drying was likely not seen as significant, and the girder’s resulting increased electrical resistance, not noted in the literature either, would not have been seen as a problem.
The Zeppelin men also took note of another lesson learned by the British rigid program: aluminum alloys corrode. Images of the “rotten” R-100 girders being broken up and sold for scrap undoubtedly inspired the LZ-129 structure’s treatment with alkyd/acrylic lacquer containing a phthalocynanine blue dye. (Photo: This was accurately formulated for priming every piece of the Zeppelin Museum’s LZ-129 section re-creation.) The fact it was still flammable after drying was likely not seen as significant, and the girder’s resulting increased electrical resistance, not noted in the literature either, would not have been seen as a problem.
Perfecting an impermeable balloon material as lightweight-sheer as possible was key to rigid airship development. Some early airship cells tried rubberized cotton; but, since rubbing it could generate sparks, an improvement was developed using bovine intestine glued on cotton. Accompanying an excellent photo of a ZR-1 cell on page 81 of Robinson and Keller’s book UP SHIP is the caption “Note the transparency and fragility of the material, made up of separate longitudinal gores of light cotton fabric, with goldbeaters’ skins shingled on.” Of the S-L airships, Karl-Heinz Hoffmann wrote, “15 gas cells provided buoyancy. The cells were made of double rubberized fabric or of goldbeater skin. Gold brocade or goldbeater skin originated from the upper skin layers of the appendices of sheep or cattle. After appropriate treatment, seven staggered layers were glued  together to form one surface from which the gas cell was made. This extremely gas-proof cell only weighed 150 g/m². However, it must be mentioned here that this material is very costly and susceptible to bacteria with a decomposing effect.” Visitors to the Nordholz Museum were shown actual cell material; in the photo, the WWI cell in the case has a bullet hole in it.
together to form one surface from which the gas cell was made. This extremely gas-proof cell only weighed 150 g/m². However, it must be mentioned here that this material is very costly and susceptible to bacteria with a decomposing effect.” Visitors to the Nordholz Museum were shown actual cell material; in the photo, the WWI cell in the case has a bullet hole in it.
The bovine intestine cell material just would not do for the giant new cells required for the LZ-129; the Zeppelin men would have to rely their American allies. It’s said the Germans grudgingly accepted the patent trade deals with Goodyear specifically to get the excellent gelatin-latex cell material, which is listed as losing less than 100 cc of gas per square meter per 24 hours. This allowed the Germans to abandon the cattle-gut shingled cotton cells, whose greater permeability leaked hydrogen at the rate of one liter per square meter every 24 hours.
 The sheer gas cells would not last long if exposed to the elements, and every Class A airship base was equipped for their special handling. (Photo: Moffett Hangar #1’s cell-storage “cork room” was exposed when the building’s cladding was deemed hazardous and removed.) Once installed and inside the fabric covered structure, the sun’s UV rays would still penetrate the cloth. So, from the equator up on LZ-129, the first coat of nitrate dope contained red iron oxide, seeking to extend cell life. Aluminum powder was added to successive coats over the entire cover, best technology available to combat superheat/cooling temperature swings to maintain a more controllable static condition.
The sheer gas cells would not last long if exposed to the elements, and every Class A airship base was equipped for their special handling. (Photo: Moffett Hangar #1’s cell-storage “cork room” was exposed when the building’s cladding was deemed hazardous and removed.) Once installed and inside the fabric covered structure, the sun’s UV rays would still penetrate the cloth. So, from the equator up on LZ-129, the first coat of nitrate dope contained red iron oxide, seeking to extend cell life. Aluminum powder was added to successive coats over the entire cover, best technology available to combat superheat/cooling temperature swings to maintain a more controllable static condition.
So while Graf’s older cells all but leaked like sieves by comparison even to the much greater square yardage of LZ-129’s cells, nonetheless LZ-129 would contain every safety design element held necessary by the Zeppelin men, and add ignition-less engines to boot. Yet, somehow surpassing the previously discussed helium-only design myth, is the more persistent misconception that LZ-129 was somehow more vulnerable to its lifting gas. It was not so. Seepage and small leaks were never a problem in LZ-129 either, since the nitrate-doped “fine Egyptian linen” outer covering was engineered to “breathe” as the ship changed altitude. It was simply incapable of containing the gas. Doped fabric is far too permeable to contain the hydrogen molecule, second smallest in the universe. These materials’ properties had long been known from experience in practice.
 Even relatively large volumes of gas escaping a damaged cell was not a problem under the fabric cover. Earlier Zeppelins and some British ships did not possess the elaborate valve-discharge isolation trunks of later LZs, some suggesting this was a S-L idea. In R.101 (photo) gas valves more than two feet in diameter vented directly under the cover. In an excerpt from “The Airmen Who Would Not Die” by John Fuller, we read of R.101: “In spite of the sturdy resistance to the storm, the pitch and the roll of the ship was considerable. Hunt, the chief helmsman, was inside the guts of the envelope watching the gas bags. He didn’t like what he saw. As he stumbled along the catwalk running the length of the ship, every gasbag from number three to number 14 was surging from side to side, and from fore to aft. In spite of the wire mesh intended to hold the bags in place, they were rubbing and chafing against the radial struts around the mainframes. Bag number 14 was getting worked over by the top burger of the ship, where several nuts and bolts were projecting. When he reached number eight, he was horrified to see that a 9 inch rip was formed by a radial struts, and the bag was becoming flabby, like a toy balloon stuck with a pin. More than half of the hydrogen was gone. On top of that, the safety valves at the base were opening up half an inch on every roll of the ship. It was easy for Hunt to see what was happening. The huge hull was actually rolling around the bags, rubbing them as raw as a blistered foot. Every bag was affected, to a greater or lesser degree. An enormous patchwork job was obviously in order. …the craft completed over 1000 miles in the air on a single 30 hour trip on November 18 they entered a fog of 750 feet deep on the way home to Cardington, but navigator Johnston brought the ship in directly over the mast.”
Even relatively large volumes of gas escaping a damaged cell was not a problem under the fabric cover. Earlier Zeppelins and some British ships did not possess the elaborate valve-discharge isolation trunks of later LZs, some suggesting this was a S-L idea. In R.101 (photo) gas valves more than two feet in diameter vented directly under the cover. In an excerpt from “The Airmen Who Would Not Die” by John Fuller, we read of R.101: “In spite of the sturdy resistance to the storm, the pitch and the roll of the ship was considerable. Hunt, the chief helmsman, was inside the guts of the envelope watching the gas bags. He didn’t like what he saw. As he stumbled along the catwalk running the length of the ship, every gasbag from number three to number 14 was surging from side to side, and from fore to aft. In spite of the wire mesh intended to hold the bags in place, they were rubbing and chafing against the radial struts around the mainframes. Bag number 14 was getting worked over by the top burger of the ship, where several nuts and bolts were projecting. When he reached number eight, he was horrified to see that a 9 inch rip was formed by a radial struts, and the bag was becoming flabby, like a toy balloon stuck with a pin. More than half of the hydrogen was gone. On top of that, the safety valves at the base were opening up half an inch on every roll of the ship. It was easy for Hunt to see what was happening. The huge hull was actually rolling around the bags, rubbing them as raw as a blistered foot. Every bag was affected, to a greater or lesser degree. An enormous patchwork job was obviously in order. …the craft completed over 1000 miles in the air on a single 30 hour trip on November 18 they entered a fog of 750 feet deep on the way home to Cardington, but navigator Johnston brought the ship in directly over the mast.”
 LZ-129 would continue the practice of using ramie cord netting to spread the delicate cell’s lift across the hard structure. Similar to the netting seen in this LZ-126 construction photo, Dr. Addison Bain also drew up this explanatory graphic to illustrate everything involved with attaching the fabric the the girderwork, and the
LZ-129 would continue the practice of using ramie cord netting to spread the delicate cell’s lift across the hard structure. Similar to the netting seen in this LZ-126 construction photo, Dr. Addison Bain also drew up this explanatory graphic to illustrate everything involved with attaching the fabric the the girderwork, and the  complex system used to stiffen and seal up its edges. Here also flammability was not a major concern, the netting also nitrate-doped for longevity. A detailed full sized mockup of a joint line showing its various elements is on display at the Zeppelin Museum. The literature does not contain any reference to tests of the covering’s conductivity, but this must have seemed somewhat irrelevant since it was sewn to the girders with non-conductive ramie cord as well.
complex system used to stiffen and seal up its edges. Here also flammability was not a major concern, the netting also nitrate-doped for longevity. A detailed full sized mockup of a joint line showing its various elements is on display at the Zeppelin Museum. The literature does not contain any reference to tests of the covering’s conductivity, but this must have seemed somewhat irrelevant since it was sewn to the girders with non-conductive ramie cord as well.
Furthermore, ever since a wartime Zeppelin suffered structural failure following an experimental sealed-up outer cover that could not quickly to equalize air pressure as the airship changed altitude, every rigid’s enclosed air space was designed to “breathe” easily. This prevented damage even when caught in the most violent updrafts, as had happened to Graf during at least one dishes-crashing, passenger-upsetting episode. This design feature had the extra added benefit of diluting larger hydrogen leaks while they made a bee-line for the stratosphere at 3 meters per second. Small wonder the expert who testified at the LZ-129 accident inquiry, Earle, specifically disputed the “trapped-under-the-cover” musing, stating he did not believe a combustible concentration could possibly form under the cover.
The Germans had long believed in isolating the output of the cell’s valves, both manual and automatic, into vent tunnel stacks. When hydrogen was valved, the “chimney effect” – noted to work even in the hangar – had even larger amounts of the pure gas venting immediately diluted with air rushing up from below. The mixture, which  if voluminous could possibly remain in combustible concentrations for some fleeting seconds, streamed out topside vent hoods. As the dictionary reminds us, burning oxy-hydrogen fire will not ignite mildly flammable materials even in close proximity. The literature does not specify the material with which the framework of the vent stacks was covered. However the material was obviously tested to withstand the brief bursts of oxy-hydrogen fire possible from conceivably combustible concentrations in the unlikely even some ignition source got into the stack. Herman Van Dyk was re-created the various components of the system in his drawing seen here. The system was put to the extreme test while returning from the airship’s first South America trip, when an automatic valve chattering at near pressure height suddenly stuck open and emptied the cell right up to the valve line. Once outside, as in that case still in combustible concentrations, the more than ample ignition found in atmospheric electricity would burn it off harmlessly without passengers or crew being aware of it. The incident proved an oxy-hydrogen burst couldn’t back up and radiate a significant amount of BTUs on the vent, which was metallic anyway.
if voluminous could possibly remain in combustible concentrations for some fleeting seconds, streamed out topside vent hoods. As the dictionary reminds us, burning oxy-hydrogen fire will not ignite mildly flammable materials even in close proximity. The literature does not specify the material with which the framework of the vent stacks was covered. However the material was obviously tested to withstand the brief bursts of oxy-hydrogen fire possible from conceivably combustible concentrations in the unlikely even some ignition source got into the stack. Herman Van Dyk was re-created the various components of the system in his drawing seen here. The system was put to the extreme test while returning from the airship’s first South America trip, when an automatic valve chattering at near pressure height suddenly stuck open and emptied the cell right up to the valve line. Once outside, as in that case still in combustible concentrations, the more than ample ignition found in atmospheric electricity would burn it off harmlessly without passengers or crew being aware of it. The incident proved an oxy-hydrogen burst couldn’t back up and radiate a significant amount of BTUs on the vent, which was metallic anyway.
Another popular misconception was that vented hydrogen, free to find oxygen in the outside atmosphere, could also find an ignition source in those fleeting instants before it dispersed, dissolved and rose away. Nitrate-doped fabric, after all, is quite flammable, so it would seem vulnerable to something as hot as burning oxy-hydrogen. Many an airplane lost its  fabric covering from most any ignition source which supplied the required ignition temperature – typically less than 200 degrees C. While intense, the fabric’s combustion is sometimes so rapid there is insufficient time for its radiant heat to ignite other materials. Pearl Harbor attack photos commonly show fabric burnt off airplane control surfaces but with the metal skins unharmed. Some accidents are known to leave gasoline tanks intact and even wooden airplane skeletons recognizable (photo). The fabric outer cover, the first thing in harm’s way, was the Achilles’ Heel of Zeppelins in combat, lighting off at the exit wound much as it did for “flaming coffin” airplanes, or most anything else so attacked, down to straw huts. The hydrogen cells inside, if torn by explosive (Brock) shells and venting into the air to be ignited by incendiary (Buckingham, Pomeroy, Sparklet) shells, would find their outer cover already ablaze. Of course, oxy-hydrogen flames are a power of ten hotter than nitrate-doped fabrics’ ignition. But vented gas haphazardly mixing with air in the presence of ignition sources (found at altitude) on its way skyward cannot radiate enough BTUs downward and backward for sufficient time to make some measurable difference in the rate the fabric was burning. Hundreds of hydrogen-filled blimps vented gas directly into the air at static-rich altitudes but their flammable envelopes shrugged off any ill effects. Nonetheless armchair analysts have suggested some “bizarre conditions” caused oxy-hydrogen to be both present and first to ignite, passing destructive flame fronts back to the flammable materials. The literature does not reveal any testing that showed such a vulnerability, but out of an abundance of caution, it was standard practice not to intentionally vent gas when firing topside machine guns or flying in a lightning-rich thunderstorm.
fabric covering from most any ignition source which supplied the required ignition temperature – typically less than 200 degrees C. While intense, the fabric’s combustion is sometimes so rapid there is insufficient time for its radiant heat to ignite other materials. Pearl Harbor attack photos commonly show fabric burnt off airplane control surfaces but with the metal skins unharmed. Some accidents are known to leave gasoline tanks intact and even wooden airplane skeletons recognizable (photo). The fabric outer cover, the first thing in harm’s way, was the Achilles’ Heel of Zeppelins in combat, lighting off at the exit wound much as it did for “flaming coffin” airplanes, or most anything else so attacked, down to straw huts. The hydrogen cells inside, if torn by explosive (Brock) shells and venting into the air to be ignited by incendiary (Buckingham, Pomeroy, Sparklet) shells, would find their outer cover already ablaze. Of course, oxy-hydrogen flames are a power of ten hotter than nitrate-doped fabrics’ ignition. But vented gas haphazardly mixing with air in the presence of ignition sources (found at altitude) on its way skyward cannot radiate enough BTUs downward and backward for sufficient time to make some measurable difference in the rate the fabric was burning. Hundreds of hydrogen-filled blimps vented gas directly into the air at static-rich altitudes but their flammable envelopes shrugged off any ill effects. Nonetheless armchair analysts have suggested some “bizarre conditions” caused oxy-hydrogen to be both present and first to ignite, passing destructive flame fronts back to the flammable materials. The literature does not reveal any testing that showed such a vulnerability, but out of an abundance of caution, it was standard practice not to intentionally vent gas when firing topside machine guns or flying in a lightning-rich thunderstorm.
 LZ-127 Graf had become the most successful airship ever, traveling the world while its operators racked up invaluable experience in the process. One challenge faced by all airships is that of the unpredictable weight of rain impacting the considerable acreage of surface area. In flight, it is normally handled with dynamic lift, changing the elevator angle back as soon as the weight evaporates. At slower air speeds there is no choice but to drop ballast, and when the weight disappears with the sun, hydrogen lift must be vented. Since the airship has a finite supply of both, very high on the “wish list” was a more water-repellent doping process. This item moved to the top of the wish list in April, 1935, following an attempted landing in Pernambuco, on which Harold Dick was along and later recalled in his book:
LZ-127 Graf had become the most successful airship ever, traveling the world while its operators racked up invaluable experience in the process. One challenge faced by all airships is that of the unpredictable weight of rain impacting the considerable acreage of surface area. In flight, it is normally handled with dynamic lift, changing the elevator angle back as soon as the weight evaporates. At slower air speeds there is no choice but to drop ballast, and when the weight disappears with the sun, hydrogen lift must be vented. Since the airship has a finite supply of both, very high on the “wish list” was a more water-repellent doping process. This item moved to the top of the wish list in April, 1935, following an attempted landing in Pernambuco, on which Harold Dick was along and later recalled in his book:
 “During the approach a dense white cloud moving in from the sea was encountered. The Graf continued through this cloud which turned out to contain no rain and was of no great consequence. With the ship down to 300 feet and closer to the field, another cloud formation similar to the first was encountered. Because the first had been of no consequence, it was assumed this would be true also of the second; the airship, at this time about 1100 pounds light, was flown through.
“During the approach a dense white cloud moving in from the sea was encountered. The Graf continued through this cloud which turned out to contain no rain and was of no great consequence. With the ship down to 300 feet and closer to the field, another cloud formation similar to the first was encountered. Because the first had been of no consequence, it was assumed this would be true also of the second; the airship, at this time about 1100 pounds light, was flown through.
“The cloud in fact contained heavy tropical rain which within a few seconds added seven tons’ weight to the ship. All available ballast, amounting to about five tons, was dropped but since the ship had little forward speed the extra heaviness could not be carried dynamically.  The Graf settled to the ground about 2000 feet short of the field, completely carrying away the lower rudder, dragging the lower fin along the ground for 300 feet before the ship came to rest. The aft engine car bounced several times on the ground, and finally when the control car struck the ground, the floor of the navigation room caved in.
The Graf settled to the ground about 2000 feet short of the field, completely carrying away the lower rudder, dragging the lower fin along the ground for 300 feet before the ship came to rest. The aft engine car bounced several times on the ground, and finally when the control car struck the ground, the floor of the navigation room caved in.
“When the Graf came to rest, the chimney of one of the native huts was sticking up into the belly of the ship. A fire was going in the stove inside the hut. It was incredible that neither the hydrogen nor the Blau gas was ignited. One of the aft engine mechanics jumped from the car, ran into the hut, and with the pot of coffee that had been cooking extinguished the fire in the stove…
“Then as rapidly as it had come, the rain stopped, the sun came out, and the Graf quickly became light. The mechanic barely made it back to his power car before the ship took off again.”
 The Herberts Company offered a solution: a better “Cellon.” To overcome these dangerous rainwater-weight swings endured with the globe-trotting Graf, their new ‘Cellon’ waterproofing was hand-lacquered over Hindenburg’s final nitrate dope layers (left). The compound was worth the money because really worked: samples of LZ-129 fabric repel water amazingly well even 80 years later, rain beading up like on a freshly waxed automotive finish.
The Herberts Company offered a solution: a better “Cellon.” To overcome these dangerous rainwater-weight swings endured with the globe-trotting Graf, their new ‘Cellon’ waterproofing was hand-lacquered over Hindenburg’s final nitrate dope layers (left). The compound was worth the money because really worked: samples of LZ-129 fabric repel water amazingly well even 80 years later, rain beading up like on a freshly waxed automotive finish.
Cellon’s success was due in part to its strongly acidic formula. Much as when an individual finds it regretful to paint lacquer over enamel, the Cellon partially dissolved the nitrate dope layers with their imbedded aluminum oxide, mixing them together. Where close to the structure, depending on the applicant’s technique, the acidic formula even reached in to melt some of the girder’s blue primer. Topside, the layers melting together included the innermost, original coat of UV-resisting iron oxide. No one realized it at the time, but mixing aluminum dust in a binder with small amounts of iron oxide yielded a chemical composition nearly identical to what would become a Polaris missile’s solid rocket motor, in which iron oxide acts as a catalyst. The whole mess glopping together had inadvertently created random, tiny blobs of thermite topside. When finally dry, the result was indeed water resistant. However, the process resulted in the final product becoming the most highly flammable and most easily ignitable airship covering in history, and having a very high electrical resistance as well. The literature does not suggest the resultant tendency to hold a charge better than any airship before was appreciated, but if so, it might have been seen as beneficial.
Zeppelin girder structures were carefully electrically bonded bow to stern, some having noted girder-melting lightning strikes passing out the tail so quickly even the fabric did not ignite. Nonetheless this exit-wound-ignition was certainly a possibility, and may even have brought down a Great War Zeppelin or two.
With the ZRS-4 design mature and construction if its rings underway, its fabric covered model (note original “arrow” fins and more forward control car) was subjected to simulated lighting strikes, reported in the local paper and preserved in the Karl Arnstein Collection, University of Akron Archives:

The final line of the caption reads, “In one case the fabric near the tail surfaces caught fire.”
The literature fails to suggest outer covers were then part of the Zeppelin’s electrical equation. With LZ-129’s new Cellon-laquered nitrate doped fabric, the covering’s newly unique electrical resistance – coupled with the isolation from the structure – would prove to be underappreciated at the time.
Following long years of construction, the Zeppelin men were ready to walk out their new airship and start making money hauling passengers and freight. Americans, a necessary part of the enterprise, would be there for the rollout, and be present for much of LZ-129’s life – and death.
Read on to The US Navy and the Hindenburg
or
Skip up to Hindenburg Götterdämmerung
or
Skip up to Hindenburg Explosion
Purchase raw video DVD The Hindenburg
back to Home Page
